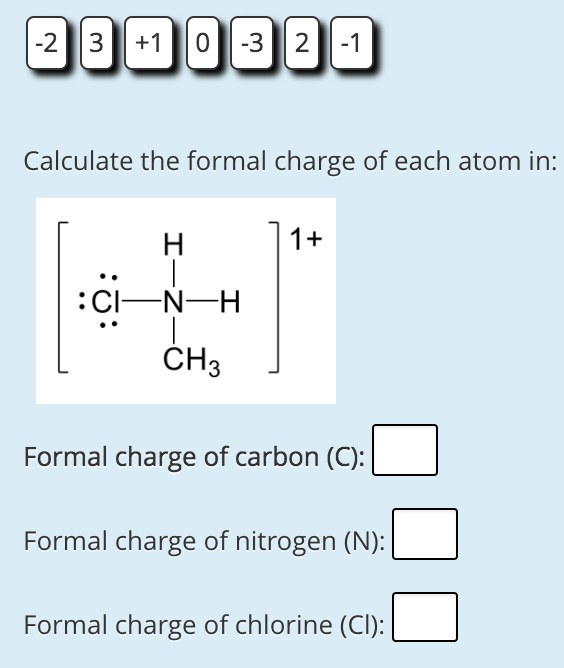
We should remember that a molecule described as a resonance hybrid never possesses an electronic structure described by either resonance form. is the number of valence electrons in the isolated. 
A more mathematical way of defining the formal charge on a particular atom is given by the. Notice that the sum of the formal charges on all the atoms equals the total charge on the whole ion. A double-headed arrow between Lewis structures indicates that they are resonance forms. electrons, and therefore a formal charge of 1. The actual electronic structure of the molecule (the average of the resonance forms) is called a resonance hybrid of the individual resonance forms. Calculating Formal Charge: The following equation determines the formal charge for each atom in a molecule or polyatomic ion. The partial charges of HgII are 0.53 and 0.55 for CH3HgSCH3 and CH3SHgIISCH3, respectively, values of about one quarter of the formal charge of Hg2+. We call the individual Lewis structures resonance forms. The actual distribution of electrons in each of the nitrogen-oxygen bonds in is the average of a double bond and a single bond. Instead, we use the concept of resonance: if two or more Lewis structures with the same arrangement of atoms can be written for a molecule or ion, the actual distribution of electrons is an average of that shown by the various Lewis structures. It is not possible to write a single Lewis structure for in which nitrogen has an octet and both bonds are equivalent. Experiments show, however, that both N–O bonds in have the same strength and length, and are identical in all other properties. We assign lone pairs of electrons to their atoms. electrons in lone pairs, or singly-occupied orbitals) and half of the electrons that it. To obtain the formal charge of an atom, we start by counting the number of valence electrons Note 1 for the neutral atom, and then subtract from it the number of electrons that it owns (i.e. A double bond between two atoms is shorter (and stronger) than a single bond between the same two atoms. We divide the bonding electron pairs equally for all ICl bonds: Step 2. Formal charge is a book-keeping formalism for assigning a charge to a specific atom. If nitrite ions do indeed contain a single and a double bond, we would expect for the two bond lengths to be different. 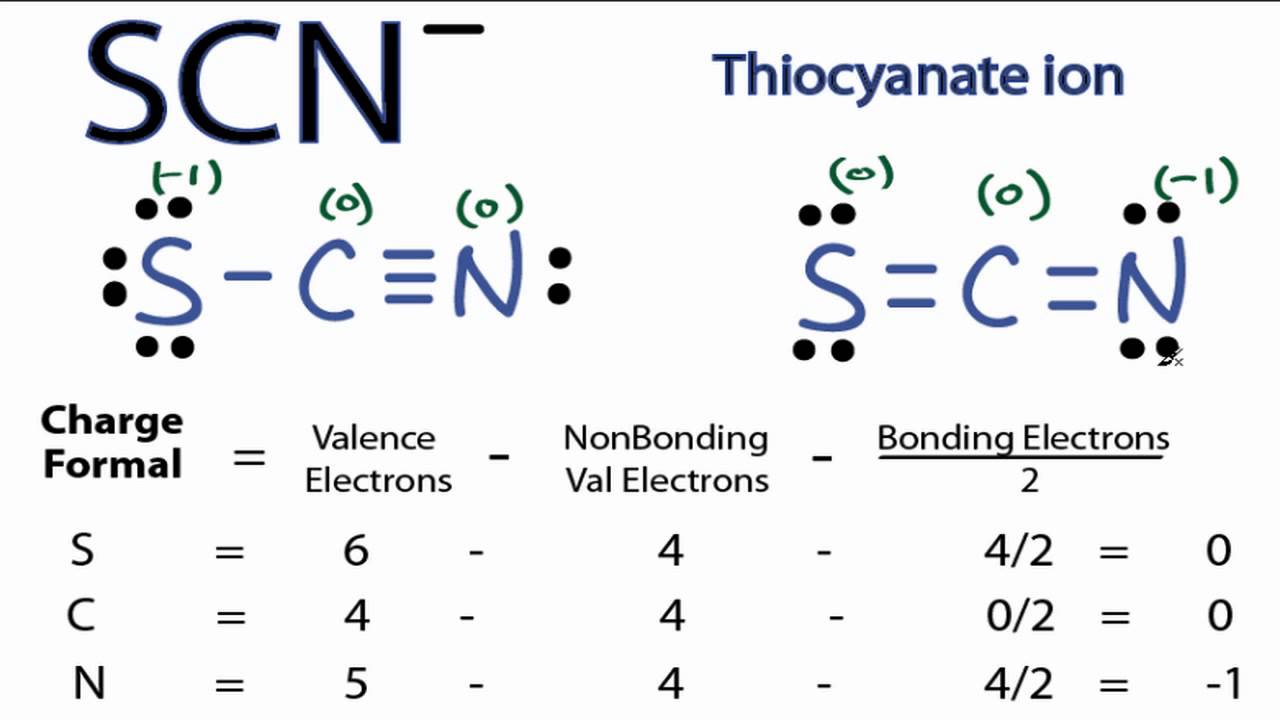
Also, it places the least electronegative atom in the center, and the negative charge on the more electronegative element (Guideline 4).\) may actually be drawn in two different ways, distinguished by the locations of the N-O and N=O bonds: 
Does formal charge determine the dipole moment Yes, of course The formal charge do determine the dipole moment. This cancels out any chance of polarity within the molecule. Thus, we calculate formal charge as follows: formal charge valence shell electrons (free atom). 2.96 Draw the most important Lewis structure for each of the following ring molecules (which have been drawn without. However, the first arrangement of atoms is preferred because it has the lowest number of atoms with nonzero formal charges (Guideline 2). The formal charges are always assumed for the perfect covalent bonds which means there is no chance of electrons to be shared unequally. Another way of saying this is that formal charge results when we take the number of valence electrons of a neutral atom, subtract the nonbonding electrons, and then subtract the number of bonds connected to that atom in the Lewis structure. Calculate the formal charges on all atoms. In the case of hemoglobin, because the carbon monoxide binds very strongly to the iron, the CO blocks the position where oxygen. When exposed to transition metal cations such as the iron in hemoglobin ( Fe 2+), the carbon is attracted to and binds to the metal. Note that the sum of the formal charges in each case is equal to the charge of the ion (–1). Formal charge can help us to understand the behavior of carbon monoxide, CO C O. All atoms in BrCl3 BrCl 3 have a formal charge of zero, and the sum of the formal charges totals zero, as it must in a neutral molecule. Lewis structures can also be useful in predicting molecular geometry in conjuntion with hybrid orbitals. Subtract this number from the number of valence electrons for the neutral atom. Possible Lewis structures and the formal charges for each of the three possible structures for the thiocyanate ion are shown here: Lewis structures, also known as Lewis-dot diagrams, show the bonding relationship between atoms of a molecule and the lone pairs of electrons in the molecule. The formal charges present in each of these molecular structures can help us pick the most likely arrangement of atoms.
0 Comments
Vimeo is full of actual humans who’d love to listen. Recapping adventures: travel videos, blogging, vlog video editorįollow us creator: show us what you #madewithvimeo.Any event: birthday card maker, wedding invitation maker, online invitation creator.Make video invitations, photo slideshows. 
Youtube intros and outros, and Youtube ads.Create Facebook posts & Facebook video covers.Instagram story editor, and story highlights maker.You can turn off auto-renew at any time from your iTunes account settings. To avoid renewal, you must disable auto-renew at least 24 hours before your subscription ends. Once your free trial ends (if any), you will be charged a subscription fee (plus any tax) per subscription period until you cancel. You can always review the total price before completing payment.ĭepending on your subscription, Vimeo Plus will renew monthly or annually, and pricing will vary by the subscription type and by country. Upon confirmation of purchase, the amount will be charged to your iTunes account. Make a video by uploading your own stickers, brand colors, custom logos, and get access to even more marketing features, available when you upgrade.Īccess to millions (yes, millions) of stock clips, hundreds of professional templates, and even more powerful video tools by making the jump to Vimeo Pro or higher. Make it move with sticker and text animations.Select a soundtrack from our licensed library.Choose a sticker and soundtrack to fit any mood.From: social ads, sales announcements and Instagram Stories, to bridal shower and birthday invitations - we’ve got you covered. Each one has been professionally designed so no matter what type of video you need, our video creation software makes it easy. You can become the video editor you always wanted to be with built-in tools and filters that make your images and video clips stand out, smooth transitions between clips, and show off a finished product that’s ready to sell. Download the Vimeo Create app to pick up where you left off and create video online with: phone, tablet, or desktop - anytime, anywhere.Įxplore over 3,000 + ready-made video templates. Vimeo Create is the best product video maker for professional-looking videos that don’t require professional editing. Start creating with our easy video maker for free and do it on any device. Create and customize videos for any occasion with our A.I. In its short time on-screen, it imparts a fair bit of additional flavour to the fall of Rapture and succeeds in answering the question of how, why, when and what Elizabeth and Booker are doing in Andrew Ryan’s neighbourhood. While it doesn’t live up to the noir billing, Burial at Sea does manage the unenviable task of tying together the universes of BioShock and BioShock Infinite. Freezing people and smashing their chilled bodies is still quite gratifying, but you’ve done it before. Anyone who’s read Chandler’s Philip Marlowe books should appreciate the writers’ ear for details like those.īurial at Sea adds one absurdly powerful new weapon that you’ll get to use for roughly fifteen or twenty minutes before the conclusion of the DLC, and renames ‘Winter Blast’ to ‘Old Man Winter,’ perhaps in the hope that some people will think it’s a new vigor/plasmid. This latter branch adds some neat little extra noir flourishes, like the hotel porter earnestly telling DeWitt to get lost before “the house dick” shows up. 
Every conversation is either about discontent at Andrew Ryan’s actions against Fontaine (it really does help to have played the first BioShock, though the ambient dialogue will fill in the basics,) Randian super-beings cruising for action, or directed at Booker himself. then right click on it - Properties - Local files - Verify. go to Library in your steam client and find bioshock infinite there. I tried reloading, changing graphics settings, but nothign helps. In the initial segment, you do indeed see this. As the chapter loads, it instantly glitching, showing my schield indefinitely breaking and me just beign dead. And, once again, he’s going to be searching for a girl: a missing child named Sally.Ī lot of pre-release hype has focused on the chance to see Rapture at its decadent heights, on New Year’s Eve 1958. That lady is Elizabeth, older, but still missing her finger. A lady sways into the office of a boozy, dishevelled private eye with a job proposition and, against his better judgement, the investigator agrees. 
The opening scenes (which you may have seen in a trailer,) however, are the classic genre intro. This first chapter slings the coat of noir around its shoulders without ever quite managing to actually put it on. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
